A Precise Synthetic Toolbox: H-Bond-Assisted Quadruple Reactivity of o-Dimethylaminoaryloximes
Mar 13, 2025·





Dr. Semyon Tsybulin
Stepan Meshalkin
Dr. Daria Tonkoglazova
Victor Bardakov
Prof. Alexander Pozharskii
Dr. Alexander Antonov
Corresponding author
·
0 min read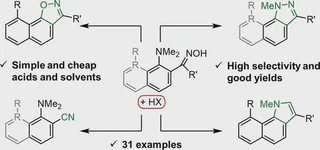
Abstract
It has been shown that azomethines, hydrazones, and oximes derived from 2(7)-carbonyl derivatives of 1,8-bis(dimethylamino)naphthalene can undergo acid-catalyzed heterocyclization leading to a nucleophilic displacement of the 1-NMe2 group. The process is believed to be directly connected with the proton sponge nature of the substrates, in which 1-NMe2, being a poor leaving group, is preliminary activated via the formation of a chelated protonated form. A number of difficult to access derivatives of benzo[g]indazole, benzo[g]quinazoline, naphtho[2,1-d]isoxazole, and 8-dimethylamino-1-naphthol have been prepared in moderate to high yields.
Publication
J. Org. Chem., 2025, 90, 4374–4381





Authors
1938-2025
Prof. Pozharskii was not only my PhD supervisor, but he was a beacon in the scientific world for me. He taught me how to properly plan an experiment, how to write research papers, how to supervise students, how to make grant applications, and how to give lectures. I am who I am today thanks to Prof. Pozharskii and his contribution to me.
