Transition-Metal-Free Synthesis of 2-Substituted Benzo[cd]Indoles via the Reaction of 1-Halo-8-lithionaphthalenes with Nitriles
May 15, 2024· ,
,
Dr. Semyon Tsybulin
M. V. Kaplanskiy
Dr. Alexander Antonov
Corresponding author
·
0 min read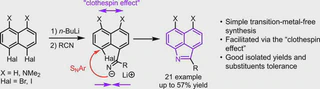
Abstract
A simple and effective organolithium approach to the synthesis of 2-substituted benzo[cd]indoles from peri-dihalonaphthalenes and nitriles has been developed. The reaction proceeds via a surprisingly easy intramolecular aromatic nucleophilic substitution facilitated by the “clothespin effect”. The discovered transformation provides good isolated yields, allows usage of an extensive range of nitriles, and demonstrates a good substituents tolerance. UV-absorption and NMR spectra of the obtained benzo[cd]indoles and their protonated forms demonstrated exclusive protonation to the indole nitrogen atom even in the presence of two NMe2 groups in positions 5 and 6 (i. e. “proton sponge” moiety).
Publication
Chem. Eur. J. 2024, 30, e202303768

