The role of conjugation in the halogen-lithium exchange: lithiation of 4,6,7,9-tetrabromo-1,3-dimethyl-2,3-dihydro-1H-perimidine
Jun 1, 2023·


Artyom Yakubenko
Prof. Elena Tupikina
Dr. Alexander Antonov
Corresponding author
·
0 min read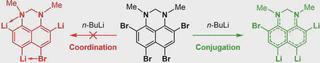
Abstract
The first case of successful suppression of the coordination of a lithium atom with a dialkylamino group by the effective conjugation of the latter with the aromatic core has been discovered. This effect controls regioselectivity of the bromine−lithium exchange in 4,6,7,9-tetrabromo-1,3-dimethyl-2,3-dihydro-1H-perimidine, which leads to products with the most effective conjugation. As a result, the product of this quadruple exchange demonstrates no tendency of the coordination of the NMe groups to neighboring lithium atoms despite the absence of steric restrictions. Experimental results are explained by means of quantum chemical calculations: geometry optimization, natural bond analysis and scans using the modredundant scheme.
Publication
Chem. Eur. J. 2023, 29, e202301439


