Spontaneous Cyclization of peri-Diiminonaphthalenes Leading to the Formation of Benzo[de]isoquinolines and Stable Benzo[de]isoquinoliniums
Feb 7, 2023·




Dmitrii Tolochenko
Dr. Semyon Tsybulin
Artyom Yakubenko
Prof. Elena Tupikina
Dr. Alexander Antonov
Corresponding author
·
0 min read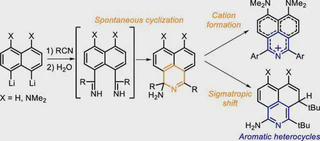
Abstract
The interaction of peri-dilithionaphthalenes with organic cyanides was studied. Instead of the expected peri-diimines, the reaction leads to the formation of three types of benzo[de]isoquinolines. Treatment of unsubstituted 1,8-dilithionaphthalene with aromatic nitriles results in the formation of 1-amino-1,3-diaryl-1H-benzo[de]isoquinolines. In contrast, 4,5-dilithio-1,8-bis(dimethylamino)naphthalene gives an aromatic isoquinolonium cation via elimination of ammonia under the same condition. Upon treatment with tert-butylcyanide, both dilithionaphthalenes undergo a transformation to 1-amino-3,4-di-tert-butyl-4H-benzo[de]isoquinolines. The observed reactivity was supported by quantum chemical calculations.
Publication
Org. Lett. 2023, 25, 977–981




