Lithiation of 2,4,5,7-tetrabromo-1,8-bis(dimethylamino)naphthalene: peculiarities of directing groups' effects and the possibility of polymetallation
Oct 25, 2021· ,
,

Artyom Yakubenko
V. V. Karpov
Prof. Elena Tupikina
Dr. Alexander Antonov
Corresponding author
·
0 min read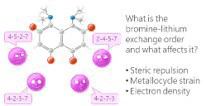
Abstract
The bromine-lithium exchange sequence upon interaction of 2,4,5,7-tetrabromo-1,8-bis(dimethylamino)naphthalene with n-BuLi was studied. Experimental results were explained by means of quantum chemical calculations. It was demonstrated that the first exchange occurs in position 4 due to the significant decrease of a steric strain of the molecule. The second exchange takes place in either position 5 or 7 due to the even more negative charge distribution in the naphthalene core. As a result, the third exchange leads to the species containing lithium in positions 2,4,5 or 2,4,7. Using a large excess of n-BuLi in hexane, 2,4,5,7-tetralithio-1,8-bis(dimethylamino)naphthalene was successfully prepared. The latter was used for the synthesis of several tetrasubstituted derivatives of 1,8-bis(dimethylamino)naphthalene by quenching with different electrophiles.
Publication
Organometallics 2021, 40, 3627–3636


