How strong is hydrogen bonding to the amide nitrogen?
Jan 18, 2020·
 ,,
,,
 ,,·
0 min read
,,·
0 min read
Dr. Vladimir Mikshiev
Corresponding author
Prof. Alexander Pozharskii
A. Filarowski
A. S. Novikov
Dr. Alexander Antonov
Prof. Peter Tolstoy
M. A. Vovk
O. V. Khoroshilova
Abstract
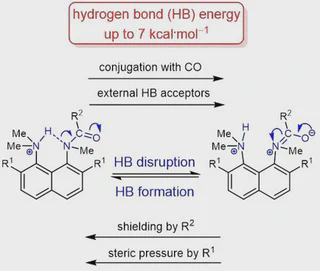
The protonation of the carboxamide nitrogen atom is an essential part of in vivo and in vitro processes (cis-trans isomerization, amides hydrolysis etc). This phenomenon is well studied in geometrically strongly distorted amides, although there is little data concerning the protonation of undistorted amides. In the latter case, the participation of amide nitrogen in hydrogen bonding (which can be regarded as the incipient state of a proton transfer process) is less well-studied. Thus, it would be a worthy goal to investigate the enthalpy of this interaction. We prepared and investigated a set of peri-substituted naphthalenes containing the protonated dimethylamino group next to the amide nitrogen atom (“amide proton sponges”), which could serve as models for the study of an intramolecular hydrogen bond with the amide nitrogen atom. X-Ray analysis, NMR spectra, basicity values as well as quantum chemical calculations revealed the existence of a hydrogen bond with the amide nitrogen, that should be attributed to the borderline between moderate and weak intramolecular hydrogen bonds (2–7 kcal ⋅ mol−1).
Publication
ChemPhysChem 2020, 21, 651–658


Authors
1938-2025
Prof. Pozharskii was not only my PhD supervisor, but he was a beacon in the scientific world for me. He taught me how to properly plan an experiment, how to write research papers, how to supervise students, how to make grant applications, and how to give lectures. I am who I am today thanks to Prof. Pozharskii and his contribution to me.
Authors
Authors


Authors
Authors