Sterically facilitated meta-lithiation of arenes, containing electron donating groups
Dec 6, 2019·

 ·
0 min read
·
0 min read
Dr. Alexander Antonov
Corresponding author
Victor Bardakov
Dr. Valeriia Mulloyarova
Abstract
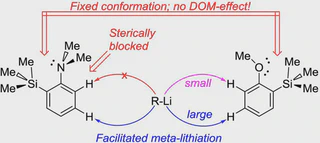
The influence of the bulky trimethylsilyl substituent on the selectivity of metallation of dimethylaniline, anisole and 1-dimethylaminonaphthalene is studied. The neighboring SiMe3 group forces dimethylamino and methoxy groups to occupy a conformation with an unshared electron pair turned towards silicon atom. This forced conformation prevents NMe2 and OMe groups from providing the DOM-effect, thus facilitating meta-metallation with the use of bulky LiCKOR. While the inverted NMe2 group completely suppresses ortho-metallation, the less bulky and more electron withdrawing OMe group demonstrates more rotation freedom allowing selective ortho-metallation with smaller reagents such as n-BuLi or tert-BuLi.
Publication
J. Organomet. Chem. 2020, 906, 121068


