Laying the way to meta-functionalization of naphthalene proton sponge via the use of Schlosser's superbase
Dec 12, 2017·

 ,,·
0 min read
,,·
0 min read
Dr. Alexander Antonov
Corresponding author
Victor Bardakov
Prof. Alexander Pozharskii
M. A. Vovk
A. D. Misharev
Abstract
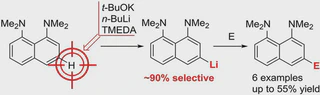
Lithiation of 1,8-bis(dimethylamino)naphthalene (DMAN) with Schlosser’s superbase (n-BuLi–t-BuOK) in the presence of TMEDA in hexane was examined. It has been shown that, compared with previously studied n-BuLi–TMEDA or t-BuLi–TMEDA mixtures, this reagent provides much more selective meta-lithiation. A variety of 3-substituted and 3,6-disubstituted derivatives of DMAN has been prepared in a good to reasonable yield after quenching the reaction mass with different electrophiles. A possibility of further functionalization of thus introduced meta-substituents to synthesize more complex 3-substituted derivatives of DMAN is also demonstrated.
Publication
J. Organomet. Chem. 2018, 855, 18–25



Authors
1938-2025
Prof. Pozharskii was not only my PhD supervisor, but he was a beacon in the scientific world for me. He taught me how to properly plan an experiment, how to write research papers, how to supervise students, how to make grant applications, and how to give lectures. I am who I am today thanks to Prof. Pozharskii and his contribution to me.
Authors
Authors