Ring lithiation of 1,8-bis(dimethylamino)naphthalene: another side of the „proton sponge coin”
Sep 9, 2015·
 ,,
,, ,·
0 min read
,·
0 min read
Dr. Alexander Antonov
Prof. Alexander Pozharskii
Corresponding author
,V. A. Ozeryanskii
A. Filarowski
K. Yu. Suponitsky
Prof. Peter Tolstoy
M. A. Vovk
Abstract
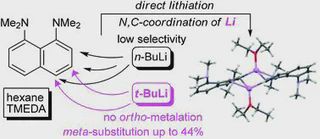
It has been found that 1,8-bis(dimethylamino)naphthalene (DMAN), unlike N,N-dimethylaniline, undergoes ring metallation in the n-BuLi–TMEDA–Et2O system with a low selectivity and in poor total yields. The situation is significantly improved in the t-BuLi–TMEDA–n-hexane system when 3- and 4-lithium derivatives become the only reaction products obtained in good yields. The formation of 3-Li-DMAN is especially desired since no method of direct meta-functionalization of DMAN is known to date. The relative stability and structure of DMAN lithium derivatives have been examined with the help of X-ray and multinuclear NMR measurements as well as DFT calculations.
Publication
Dalton Trans. 2016, 44, 17756–17766


Authors
1938-2025
Prof. Pozharskii was not only my PhD supervisor, but he was a beacon in the scientific world for me. He taught me how to properly plan an experiment, how to write research papers, how to supervise students, how to make grant applications, and how to give lectures. I am who I am today thanks to Prof. Pozharskii and his contribution to me.
Authors
Authors
Authors

Authors