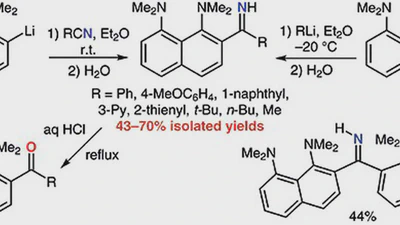
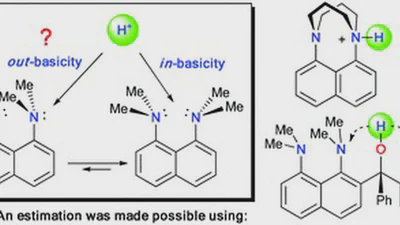
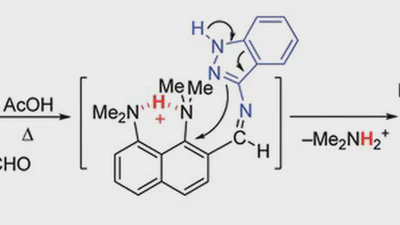
Ring lithiation of 1,8-bis(dimethylamino)naphthalene: another side of the „proton sponge coin”
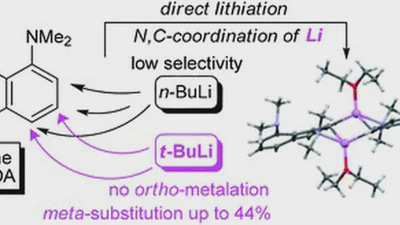
It has been found that 1,8-bis(dimethylamino)naphthalene (DMAN), unlike N,N-dimethylaniline, undergoes ring metallation in the n-BuLi–TMEDA–Et2O system with a low selectivity and …

![Base-promoted transformation of 2-C(O)R-1,8-bis(dimethylamino)naphthalenes into benzo[g]indole derivatives featured image](/publications/04/publications/04/featured_hu_21d067fcb054fe1c.webp)