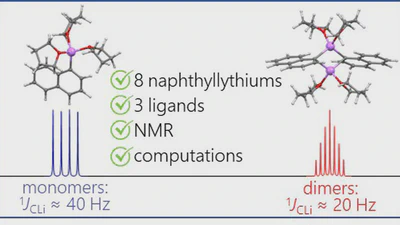
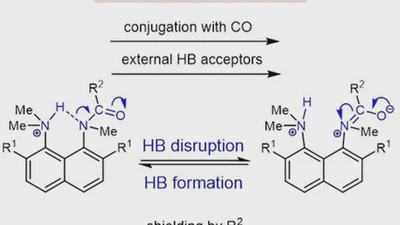
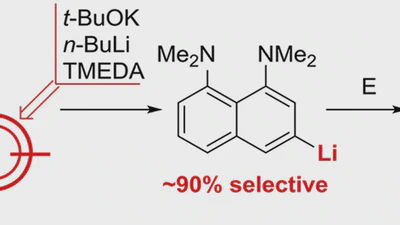
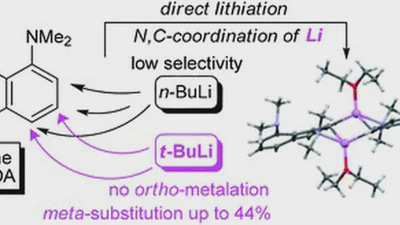
Aggregation behaviour of lithionaphthalenes in solution: experimental and theoretical study
The aggregation of a series of mono- and dilithionaphthalenes in THF solutions in the presence of tetramethylethylenediamine (TMEDA) and pentamethyldiethylenetriamine (PMDTA) was …