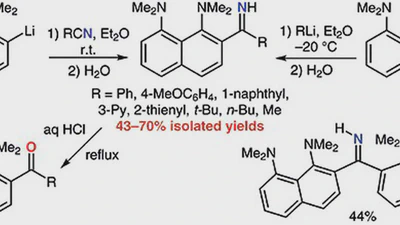
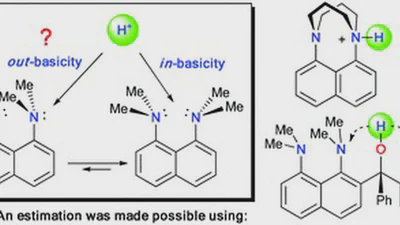
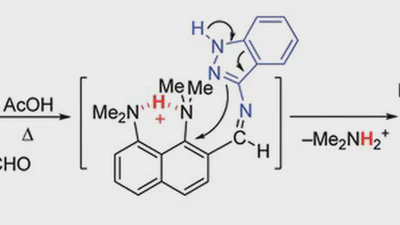
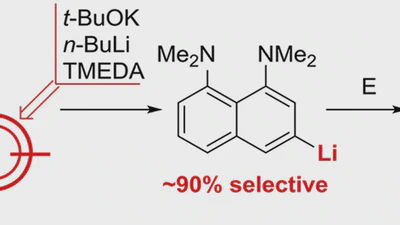
1,8-Bis(dimethylamino)naphthyl-2-ketimines: Inside vs outside protonation
The structure and protonation behaviour of four ortho-arylketimines of 1,8-bis(dimethylamonio)naphthalene with a different number of methoxy groups in an aromatic substituent were …

![Reaction of 2-Trifluoroacetyl-1,8-Bis(dimethylamino)naphthalene with Strong Organic Bases: Acidic Ionization of 1-NMe2 Group Resulting in the Formation of Benzo[g]indole Derivatives versus Nucleophilic Addition to C=O Group featured image](/publications/08/publications/08/featured_hu_91ae6f6dd030bd67.webp)
![10-Dimethylamino derivatives of benzo[h]quinoline and benzo[h]quinazolines: fluorescent proton sponge analogues with opposed peri-NMe2/–N= groups. How to distinguish between proton sponges and pseudo-proton sponges featured image](/publications/07/publications/07/featured_hu_520cda48527b48d8.webp)
![Tandem synthesis of 10- dimethylaminobenzo[h]quinazolines from 2-ketimino-1,8-bis(dimethylamino)naphthalenes via nucleophilic replacement of the unactivated aromatic NMe2 group featured image](/publications/06/publications/06/featured_hu_effab4874e9d2526.webp)
![Base-promoted transformation of 2-C(O)R-1,8-bis(dimethylamino)naphthalenes into benzo[g]indole derivatives featured image](/publications/04/publications/04/featured_hu_21d067fcb054fe1c.webp)