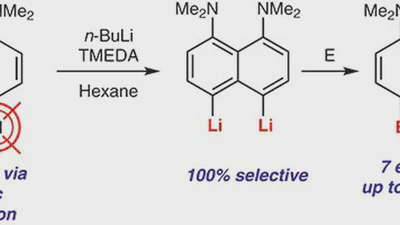
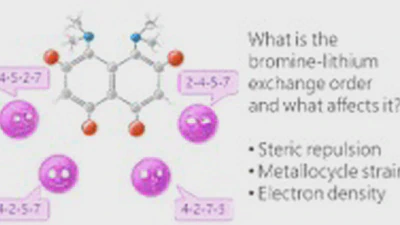
Lithiation of 2,4,5,7-tetrabromo-1,8-bis(dimethylamino)naphthalene: peculiarities of directing groups' effects and the possibility of polymetallation
The bromine-lithium exchange sequence upon interaction of 2,4,5,7-tetrabromo-1,8-bis(dimethylamino)naphthalene with n-BuLi was studied. Experimental results were explained by means …

![Proton-induced fluorescence in modified quino[7,8-h]quinolines: dual sensing for protons and π-donors featured image](/publications/11/publications/11/featured_hu_4ba2cc4c963068d8.webp)