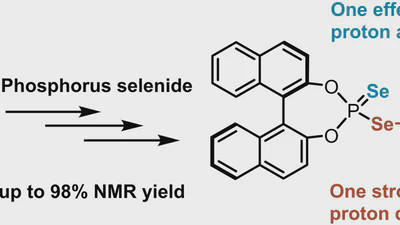
Chiral Diselenophosphoric Acids for Ion Pair Catalysis: A Novel Approach to Enhance Both Proton Donating and Proton Accepting Properties
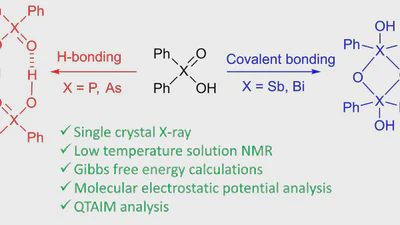
The activation of poorly reactive substrates via strong chiral acids is a central topic in asymmetric ion pair catalysis these days. Despite highly successful scaffolds such as …


![Spontaneous Cyclization of peri-Diiminonaphthalenes Leading to the Formation of Benzo[de]isoquinolines and Stable Benzo[de]isoquinoliniums featured image](/publications/26/publications/26/featured_hu_8e0754dad17c394b.webp)
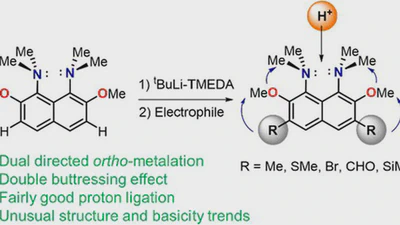
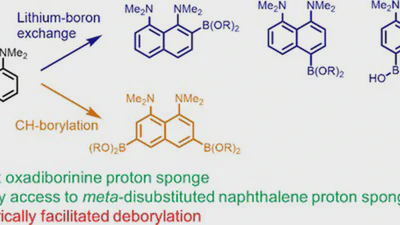
![peri-Interactions in 1,8-bis(dimethylamino)naphthalene ortho-ketimine cations facilitate [1,5]-hydride shift: selective synthesis of 1,2,3,4-tetrahydrobenzo[h]quinazolines featured image](/publications/22/publications/22/featured_hu_e06f2e83fe081219.webp)